States of Matter
8.MS-PS1-4. Develop a model that describes and predicts changes in particle motion, relative spatial arrangement, temperature, and state of a pure substance when thermal energy is added or removed.
· Emphasis is on qualitative molecular-level models of solids, liquids, and gases to show that adding or removing thermal energy increases or decreases kinetic energy of the particles until a change of state occurs.
· Emphasis is on qualitative molecular-level models of solids, liquids, and gases to show that adding or removing thermal energy increases or decreases kinetic energy of the particles until a change of state occurs.
Temperature v.s Heat and
Absolute Zero
|
Kinetic Energy
Gizmo Question guide
Temperature vs. Heat
|
Temperature and Absolute Zero
|
|
Plasma
- The 4th state of matter. |
|
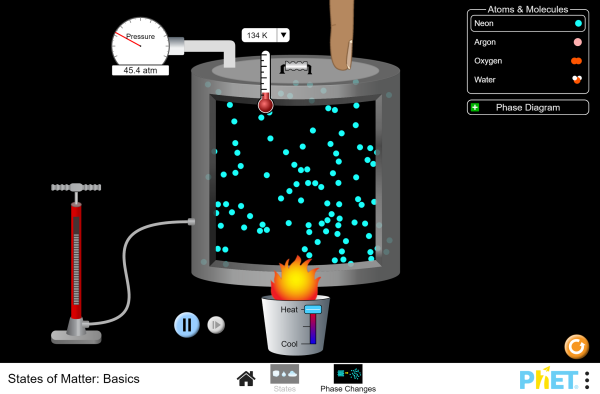
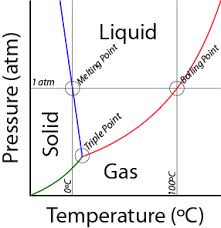
Pressure affects B.P. as well as temperature.
Charles and Boyles Law |
|
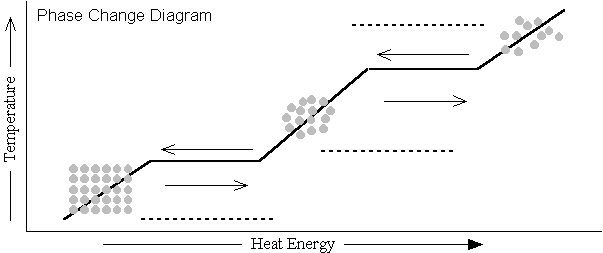
Time/Temp Graphs |
|